Pesquisa otimiza controle biológico de praga que afeta cultura de soja
Cientistas da Unesp e da Universidade Estadual de Oklahoma verificaram em campo que liberação ideal de vespa que neutraliza o percevejo-marrom deve ser realizada de 30 em 30 metros
Pesquisa demonstra relação entre poluição e riscos cardíacos em moradores de São Paulo
Estudo da USP, publicado na revista Environmental Research, analisou resultado das autópsias de 238 pessoas e dados epidemiológicos; perigo é maior para hipertensos
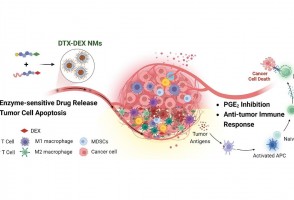
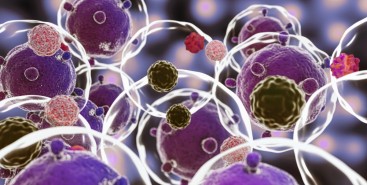
Cientistas do Brasil e da Índia criam tratamento promissor contra tumores sólidos
Em testes com animais, nanopartículas contendo substâncias já aprovadas para uso humano reduziram a inflamação no microambiente biológico em que cânceres desse tipo se instalam e vicejam, facilitando a ação do sistema imune

Estão abertas as inscrições para a 3ª edição do Prêmio Ciência para Todos
Estudantes e educadores da rede pública paulista têm até 3 de junho para se inscrever no edital, que aceita projetos de pesquisa em qualquer área do conhecimento envolvendo métodos da ciência para solucionar problemas
Cientistas brasileiros vão buscar na Romênia os traços dos últimos neandertais
A missão, liderada pelo arqueólogo e antropólogo Walter Neves, objetiva entender como se deu o contato entre os Sapiens e os Neandertais. E por que estes desapareceram
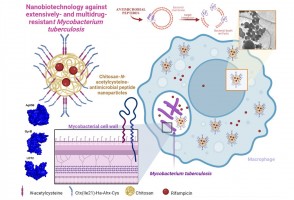
Cocaína é contaminante emergente preocupante na baía de Santos, afirma pesquisador
Droga se acumula não só na água, mas em sedimentos e organismos marinhos e representa alto risco ecológico, apontou o professor da Unifesp Camilo Seabra durante a FAPESP Week Illinois
Escola São Paulo de Ciência Avançada apresenta abordagem inovadora para tratar questões ambientais
Trabalho com foco na transdisciplinaridade busca a participação dos diversos atores sociais para enfrentar as mudanças globais. Tema foi destaque em evento realizado este mês em São Luiz do Paraitinga
OPORTUNIDADES FAPESP
Doutorado em biofísica
EPM-Unifesp
Inscrições até 30/04/2024
TT-3 em imunologia e biologia molecular
FMRP-USP
Inscrições até 30/04/2024
PD em epidemiologia e inteligência artificial
UFMG
Inscrições até 30/04/2024
PD em medicina
EPM-Unifesp
Inscrições até 30/04/2024
PD em dinâmica de fluidos e ciência dos materiais
RCGI/Poli-USP
Inscrições até 30/04/2024
Estão abertas as inscrições para a 3ª edição do Prêmio Ciência para Todos
Cientistas brasileiros vão buscar na Romênia os traços dos últimos neandertais
Pesquisa demonstra relação entre poluição e riscos cardíacos em moradores de São Paulo
Escola São Paulo de Ciência Avançada apresenta abordagem inovadora para tratar questões ambientais
Terceira Conferência FAPESP de 2024 aborda a formação de médicos-cientistas
Chamada com UE apoiará pesquisas em ciência e engenharia de materiais
Livro destaca a relevância das mulheres na construção do pensamento geográfico
Capítulo 7: Expedição termina com mais de 130 espécies de peixes coletadas

Último episódio da série de reportagens faz um balanço sobre a viagem de um grupo de pesquisadores do Museu de Zoologia da USP que, ao longo de duas semanas, percorreu os rios Negro, Preto e Jauaperi, nos Estados do Amazonas e de Roraima
Capítulo 6: Viagem ao rio Negro marcou reencontro após expedições pioneiras nos anos 1990

Uma das artes de pesca utilizadas para coletar peixes-elétricos na Expedição DEGy Rio Negro foi empregada pela primeira vez em larga escala em água doce no projeto Calhamazon, que reuniu pesquisadores do Brasil e dos Estados Unidos entre 1993 e 1996
Capítulo 5: Acidente com raia não atrapalha o ritmo das coletas no Jauaperi

Baixa gravidade da lesão, atendimento médico rápido e cuidados adequados fizeram com que pesquisador pudesse voltar aos trabalhos no mesmo dia em que foi ferroado por peixe peçonhento. Na Amazônia, casos muitas vezes se agravam por carência de assistência especializada
Vídeos
Diário de Campo - Rio Negro
O Legado Suíço-Brasileiro na Amazônia: Arte, Ciência e Sustentabilidade
02/03/2024 a 30/04/2024
Introdução à programação com Python para alunas do ensino médio ou concluintes
30/03/2024 a 27/04/2024
Minicursos para estudantes do ensino médio 2024
06/04/2024 a 08/06/2024
Treinamento de plataformas e bases de dados científicas
09/04/2024 a 30/04/2024
14ª Semana da Computação
22/04/2024 a 26/04/2024
20º Congresso Brasileiro de Língua Portuguesa
25/04/2024 a 27/04/2024
Pulverização de lavouras vai ganhar aeronave especializada
Startup apoiada pelo PIPE-FAPESP está desenvolvendo helicóptero autônomo para pulverizar plantações em terreno íngreme
Teste irá permitir detecção pouco invasiva de micrometástases
Tecnologia pode possibilitar produção de enzimas no Brasil
Pulverização de lavouras vai ganhar aeronave especializada
Startup apoiada pelo PIPE-FAPESP está desenvolvendo helicóptero autônomo para pulverizar plantações em terreno íngreme
Chamadas FAPESP
Comunicar Ciência
Prazo: 22/01
Belmont Forum Climate, Environment, and Health
Prazo: Jan 2024
PIPE Start FAPESP-Sebrae: iniciando a jornada empreendedora de base tecnológica
Prazo: 18/03
Centros de Pesquisa em Inteligência Artificial Aplicada à Saúde
Prazo: 18/03
Fundação Nacional de Ciência da Suíça
Prazo: 22/03
Apoio a pesquisa em citricultura
Prazo: 31/03
Expedições Científicas Amazônia+10
Prazo: 29/04