


Proteome and transcriptome analyses unveil the molecular mechanisms triggered by viral infection in neural progenitor cells, the precursors of brain cells
Proteome and transcriptome analyses unveil the molecular mechanisms triggered by viral infection in neural progenitor cells, the precursors of brain cells.
Proteome and transcriptome analyses unveil the molecular mechanisms triggered by viral infection in neural progenitor cells, the precursors of brain cells.
Proteome and transcriptome analyses unveil the molecular mechanisms triggered by viral infection in neural progenitor cells, the precursors of brain cells
By Karina Toledo | Agência FAPESP – Recent research has shown that neural progenitor cells – a type of stem cell capable of differentiating into neurons and glial cells – are the preferred targets of Zika virus when it infects the brains of babies in utero.
A new study published by a consortium of Brazilian researchers unveils the molecular mechanisms that are activated during viral infection and eventually lead to the death of neural progenitor cells.
The findings offer scientists a path to follow in the search for drugs capable of avoiding or minimizing the damage to the central nervous system caused by the virus.
“We already knew infection by Zika virus causes the death of neural progenitors more than other types of cell. Now we’ve shown how they die. The data points to genomic instability and interruption of the cell cycle due to replication of the virus. The cells are no longer able to proliferate or start the differentiation process, and this gives rise to apoptosis, which is programmed cell death,” said Juliana Minardi Nascimento, a postdoctoral fellow affiliated with the Neuroproteomics Laboratory at the University of Campinas’s Biology Institute (IB-UNICAMP) in São Paulo State and a co-author of the published paper.
The conclusions are based on proteome and transcriptome analyses performed in vitro on three-dimensional cultures of neural progenitor cells. The proteome is the entire set of proteins expressed by a cell. The transcriptome is the full range of messenger RNA molecules expressed.
“Instead of culturing the cells on conventional glass plates, we grew them under rotation in highly specific conditions so that they acquired a three-dimensional shape and formed neurospheres,” Nascimento said. “Neurosphere formation marks the onset of neurogenesis, the cell differentiation process whereby new neurons are born from neural progenitor cells in the brain.”
Nascimento has a scholarship from FAPESP for her project, “Quantitative proteomics in neural cell lines and organoids derived from induced pluripotent stem cells from schizophrenia patients”.
The advantage of this model is that it more accurately mimics the functioning of these cells in a living organism. “In a two-dimensional culture each cell interacts only with its immediate neighbor, whereas in the 3D model communication takes place with all surrounding cells. The model closely resembles the brain of an embryo at a rudimentary stage of development,” she explained.
Before embarking on the procedure to create neurospheres, the researchers infected some of the cells with Zika virus isolated from the blood of a patient from Espírito Santo State, Brazil.
“The cultured cells remained in contact with the virus for two hours. It was then inactivated, and the neurosphere induction process began,” Nascimento said. “The infected cells did group into spheres but failed to progress substantially. After six days the neurospheres began to shrink, and after 12 days practically nothing was left of the infected cultures.”
On the third day after infection, the researchers observed that the cells exposed to Zika virus had grown 40% less than those in the control group. Proteomic analysis showed that 458 proteins were altered; 199 of these proteins were expressed at lower levels than in the control cells, and 259 were expressed at higher levels.
“Many translation proteins, which translate messenger RNA to form new proteins, were altered,” Nascimento said. “This is common in viral infections, as this kind of pathogen uses the cell’s machinery to produce its own proteins. We also observed an increase in the expression of molecules associated with DNA repair, such as BRCA1, indicating that viral replication was creating errors in the genetic material and making the cell unstable.”
However, what most drew the researchers’ attention was a change in the protein expression profile associated with the cell cycle.
“The results show that the cell cycle was interrupted very early on, before the cell began duplicating its genetic material,” Nascimento said. “We also found that this pause wasn’t due to the onset of differentiation, since expression of the proteins associated with neurogenesis decreased compared with the control neurospheres.”
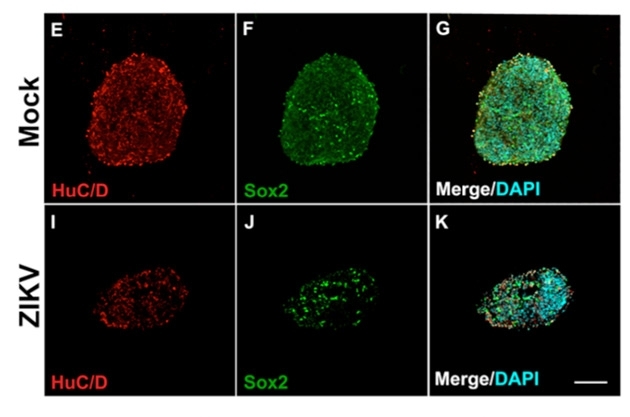
On the sixth day after infection, the population of control neurospheres was 50% larger than that of the neurospheres exposed to the virus. Non-infected cells had also advanced much farther in the process of differentiation into neurons, as evidenced by the larger number of proteins that function as specific markers for this type of cell.
“In the neurospheres exposed to Zika, the number of markers associated with neurons was much smaller. On the other hand, we observed the presence of activated caspase-3, a protein that indicates apoptosis. Another indicator of cell death was the presence of highly condensed nuclei,” Nascimento said.
Analysis performed on the 12th day after infection showed that the number of infected neurospheres was only a tenth of the number of control neurospheres. “In the case of in vitro experiments, about 21 days may be needed to obtain a functional young neuron from a neural progenitor, but in this study we show that most of the progenitors infected by Zika didn’t even last 12 days. Cell death was very fast,” she added.
Collaborations
In vitro experiments were conducted in Rio de Janeiro and coordinated by Patrícia Garcez and Stevens Rehen, researchers affiliated with the Federal University of Rio de Janeiro (UFRJ) and the D’Or Institute for Research and Education (IDOR).
Proteome analysis was performed at UNICAMP by Nascimento and Juliana Silva Cassoli, also a postdoctoral fellow, under the supervision of Professor Daniel Martins de Souza. Transcriptome assays were performed in Belém, Pará State, by collaborators at Instituto Evandro Chagas. The findings have been submitted for assessment by a high-impact journal and were posted online before completion of the peer review process in light of the subject’s importance to public health.
A preliminary draft of the article “Combined proteome and transcriptome analyses reveal that Zika virus circulating in Brazil alters cell cycle and neurogenic programs in human neurospheres” (doi.org/10.7287/peerj.preprints.2033v1) can be read at peerj.com/preprints/2033/.
Republish
The Agency FAPESP licenses news via Creative Commons (CC-BY-NC-ND) so that they can be republished free of charge and in a simple way by other digital or printed vehicles. Agência FAPESP must be credited as the source of the content being republished and the name of the reporter (if any) must be attributed. Using the HMTL button below allows compliance with these rules, detailed in Digital Republishing Policy FAPESP.





