

Scientists at the Center for Research on Redox Processes in Biomedicine (Redoxome) show that an enzyme capable of ridding cells of an oxidizing substance can be found in different parts of the mitochondrion (image: mitochondrion structure, adapted from Wikimedia Commons/Jornal da USP)
Scientists at the Center for Research on Redox Processes in Biomedicine (Redoxome) show that an enzyme capable of ridding cells of an oxidizing substance can be found in different parts of the mitochondrion.
Scientists at the Center for Research on Redox Processes in Biomedicine (Redoxome) show that an enzyme capable of ridding cells of an oxidizing substance can be found in different parts of the mitochondrion.
Scientists at the Center for Research on Redox Processes in Biomedicine (Redoxome) show that an enzyme capable of ridding cells of an oxidizing substance can be found in different parts of the mitochondrion (image: mitochondrion structure, adapted from Wikimedia Commons/Jornal da USP)
By Karina Toledo | Agência FAPESP – Cellular respiration, a process that takes place in mitochondria and guarantees energy for the organism to function, may generate byproducts known as reactive oxygen species (ROS) that are toxic for cells.
When amounts of these oxidizing substances are excessive, they interact with proteins, lipids, carbohydrates and nucleic acids to such an extent that the macromolecules concerned stop working properly. This process can lead to cell death, and in more complex organisms including humans it can contribute to the development of such diseases as cancer, arthritis, atherosclerosis, Parkinson’s and Alzheimer’s.
However, recent studies have shown that reactive molecules such as hydrogen peroxide, when present in adequate amounts, act as signaling agents in key cellular processes and thereby contribute to the proper functioning of the organism. Anti-oxidant enzymes are responsible for keeping ROS at acceptable levels and protecting cells from oxidative damage.
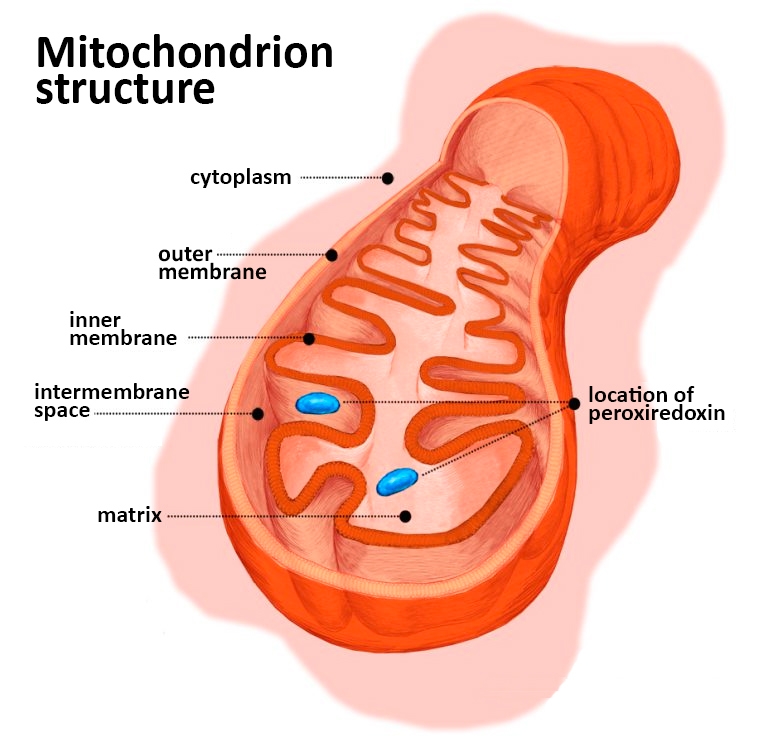
“Peroxiredoxin is the main enzyme responsible for removing hydrogen peroxide from inside mitochondria. Thus, it was known to be present in these organelles, but its exact location was unknown. Our group showed the protein can be found in two mitochondrial subcompartments, and this offers clues to the roles it plays in mitochondria,” said Luis Eduardo Soares Netto, a professor at the University of São Paulo’s Bioscience Institute (IB-USP) in Brazil.
The investigation was conducted during Fernando Gomes’s PhD research supervised by Netto, and with collaboration from Mario Barros, a professor at the university’s Biomedical Science Institute (ICB-USP) and a specialist in mitochondrial biogenesis. The group are linked to the Center for Research on Redox Processes in Biomedicine (Redoxome), one of FAPESP’S Research, Innovation and Dissemination Centers (RIDCs).
In addition to mapping the location of peroxiredoxins in mitochondria, the researchers also identified the proteins responsible for transporting this enzyme from the cellular cytoplasm where it is produced to the mitochondrial subcompartments where it must act. As a model, they used cells of brewer’s yeast, Saccharomyces cerevisiae.
The results of the study were published in the Journal of Biological Chemistry (JBC).
“Mitochondria have four subcompartments: the matrix, which is the largest, most central portion, where mitochondrial DNA is located and where the Krebs cycle [an important stage of aerobic cellular respiration] takes place; the inner membrane, where ATP [adenosine triphosphate, the energy storage molecule] is produced; the outer membrane, which separates the mitochondrion from the rest of the cell; and the intermembrane space,” Netto explained.
Each subcompartment typically contains a specific set of proteins, he went on. Peroxiredoxin is one of the few cases in which the same protein can be found at the same time in more than one place in a mitochondrion – the matrix and the intermembrane space.
“A plausible hypothesis is that its role in the matrix is to protect mitochondrial DNA against oxidative damage, in line with a more classical view of the role of anti-oxidant enzymes,” Netto said. “In the intermembrane space, it may have a regulatory function and participate in the formation of disulfide bridges between proteins.”
This kind of chemical bond, which occurs between cysteine amino acids, makes proteins more stable and in some cases may be essential for the molecule to perform properly.
“A prerequisite for disulfide bridge formation is oxidation of the target protein, and peroxiredoxin may regulate this process of electron transfer within the intermembrane space. But this needs further investigation,” Netto said.
One of the group’s future projects, he added, is to develop modified yeast expressing only the isoform of peroxiredoxin present in the matrix or only the isoform found in the intermembrane space. “That will enable us to analyze individually each of this enzyme’s functions in the mitochondrion,” he said.
Mechanism conserved
In their study of yeast mitochondria, the Redoxome researchers found that peroxiredoxin is first transported from the cell’s cytoplasm into the mitochondrion by a protein called TOM. When it reaches the inner membrane, the protein TIM23 takes over the job of bearing it to the matrix. In this subcompartment, the enzyme is processed by two proteases (enzymes that catalyze the breakdown of peptide bonds in proteins) called MPP and Oct1, which cut off pieces of the molecule and make it remain there.
In some cases, however, before TIM23 can transport the peroxiredoxin to the matrix, two other proteases found in the inner membrane – Imp1 and Imp2 – process the enzyme so that it stops midway and is then released in the intermembrane space.
“These two events are alternatives. We know that the point at which it’s determined whether peroxiredoxin will go to the matrix or remain in the intermembrane space is when it interacts with TIM23, but we aren’t sure about the context for this,” Netto said.
In one of their experiments, the group used yeast that had been genetically modified to express human peroxiredoxin and found that the enzyme was recognized by the same transporter proteins. The entire process occurred in a similar manner to that in “wild-type” (unmodified) yeast.
“The fact that the process of peroxiredoxin transport into mitochondria has been conserved throughout evolution suggests it must be important – and indeed crucial to the functioning of mitochondria,” Netto said.
Moreover, he added, since any event that affects mitochondrial functioning can jeopardize cell survival, peroxiredoxin or some of the molecules that transport it into mitochondria could be targets for the treatment of neurodegenerative diseases, cancer, diabetes, and many others.
The article “Proteolytic cleavage by the IMP complex or Oct1 peptidase controls the localization of the yeast peroxiredoxin Prx1 to distinct mitochondrial compartments” (doi: 10.1074/jbc.M117.788588) can be read at: jbc.org/content/early/2017/08/18/jbc.M117.788588.short.
Republish
The Agency FAPESP licenses news via Creative Commons (CC-BY-NC-ND) so that they can be republished free of charge and in a simple way by other digital or printed vehicles. Agência FAPESP must be credited as the source of the content being republished and the name of the reporter (if any) must be attributed. Using the HMTL button below allows compliance with these rules, detailed in Digital Republishing Policy FAPESP.





