


Researchers have identified the lipid accumulation mechanism in oocytes. Their results may help improve production in the market led worldwide by Brazil (image: release)
Researchers have identified the lipid accumulation mechanism in oocytes. Their results may help improve production in the market led worldwide by Brazil.
Researchers have identified the lipid accumulation mechanism in oocytes. Their results may help improve production in the market led worldwide by Brazil.
Researchers have identified the lipid accumulation mechanism in oocytes. Their results may help improve production in the market led worldwide by Brazil (image: release)
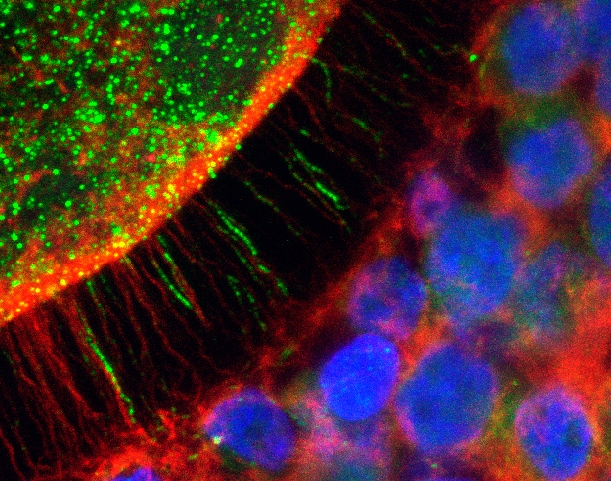
By Maria Fernanda Ziegler | Agência FAPESP – A longstanding obstacle to the market for bovine embryos is about to be removed. Researchers at the University of São Paulo’s Animal Science & Food Engineering School (FZEA-USP), in Brazil, have described a hitherto unknown mechanism of lipid accumulation in oocytes that limits the success of in vitro production of bovine embryos.
Lipid accumulation is a frequent abnormality when immature female gametes (oocytes) are used to produce embryos in vitro. This condition makes embryos less tolerant of freezing and thawing, resulting in fewer pregnancies after the embryos are transferred to receiving females.
“There are stockpiles of fat both in oocytes that develop naturally in animals’ ovaries and in those matured in incubators in the lab. However, excessive amounts of fat build up during in vitro maturation, and this is associated with reduced fertility because oocytes with large amounts of fat are less able to develop,” said Felipe Perecin, a professor at FZEA-USP and one of the authors of the study published in Scientific Reports. The research was supported by FAPESP.
Brazil is responsible for about 67% of the bovine embryos produced in vitro worldwide, according to the 2015 report of the International Embryo Technology Society (IETS). Not by chance, 214,886 (79.6%) of the 269,353 bovine embryos transferred in Brazil in 2015 were not frozen and stored but were transferred fresh after in vitro production.
“This is a problem for the bovine embryo industry in Brazil and around the world, requiring special logistics for embryo transfer in a very short period and without the possibility of freezing,” Perecin said.
The viability of embryos with excess lipid accumulation is low. “Either they’re discarded after thawing, or they’re transferred and don’t result in pregnancy, which is even worse because of the hormone protocol to synchronize the animals,” he explained.
Different flow
The researchers at FZEA-USP tracked oocytes in three situations: immature, matured in vivo, and matured in vitro. In all three cases, they identified the presence of fatty acid binding protein 3, FABP3.
“In oocytes matured in vitro or in vivo, levels of this protein were more or less the same, but when we observed the cumulus cells surrounding the oocytes, we found much higher levels of FABP3 in the in vitro system,” Perecin said.
The group then decided to investigate whether the protein transported lipids from outside into the oocyte. “This protein normally transports lipids to be oxidized in the mitochondria inside cells,” Perecin said. “We proposed the following hypothesis: Could the protein FABP3 transport lipids into the oocyte from outside, in a different flow from the classical direction?”
The investigation showed that the explanation is probably metabolic dysfunction not of the oocyte itself but of the surrounding cells and that this cell imbalance triggers an increase in the flow of lipids into the oocyte from outside.
“The end result is this accumulation of lipids, which impairs the ovum’s development potential,” Perecin said.
“We showed that in the maturation process, which lasts 24 hours, when the oocyte is removed from the female’s ovary, there is metabolic dysregulation in the surrounding cells, and this causes lipid accumulation. We found this mechanism to be very intense in the first nine hours, with a substantial flow between cumulus cells and the oocyte. Only after these nine hours do the ‘bridges’ between them, known as transzonal projections, begin to dissolve.”
The researchers plan to investigate the causes of this mechanism and find a way to block FABP3 in an attempt to reverse the system.
Perecin believes the mechanism studied in bovines may also exist in humans, although a series of studies will have to be performed to confirm this possibility.
“We’ve started another line of research,” he said. “We’re investigating whether the oocyte lipid accumulation mechanism described during in vitro maturation is also present in obesity. We think cumulus cell metabolic dysregulation may resemble the oocyte lipid accumulation associated with obesity.”
The article “Fatty acid binding protein 3 and transzonal projections are involved in lipid accumulation during in vitro maturation of bovine oocytes” (doi:10.1038/s41598-017-02467-9) by Maite del Collado, Juliano Coelho da Silveira, Juliano Rodrigues Sangalli, Gabriella Mamede Andrade, Letícia Rabello da Silva Sousa, Luciano Andrade Silva, Flávio Vieira Meirelles and Felipe Perecin can be read at nature.com/articles/s41598-017-02467-9.
Republish
The Agency FAPESP licenses news via Creative Commons (CC-BY-NC-ND) so that they can be republished free of charge and in a simple way by other digital or printed vehicles. Agência FAPESP must be credited as the source of the content being republished and the name of the reporter (if any) must be attributed. Using the HMTL button below allows compliance with these rules, detailed in Digital Republishing Policy FAPESP.





