


According to the Brazilian Society of Dermatology, atopic dermatitis affects between 15% and 25% of children and around 7% of adults in Brazil, with incidence increasing in recent years (image: Ministry of Health via CTS-CEVAP)
Drug being tested at a FAPESP’s Science Center for Development is based on a monoclonal antibody capable of combating atopic dermatitis and other types of dryness, flaking, and itching.
Drug being tested at a FAPESP’s Science Center for Development is based on a monoclonal antibody capable of combating atopic dermatitis and other types of dryness, flaking, and itching.
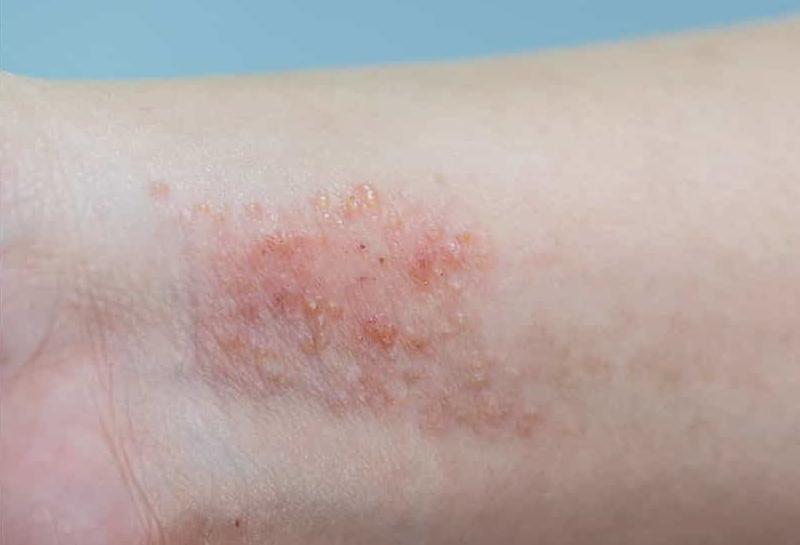
According to the Brazilian Society of Dermatology, atopic dermatitis affects between 15% and 25% of children and around 7% of adults in Brazil, with incidence increasing in recent years (image: Ministry of Health via CTS-CEVAP)
Agência FAPESP* – Researchers affiliated with the Center for Translational Science and Biopharmaceutical Development (CTS), a FAPESP Science Center for Development (SCD), have developed a monoclonal antibody capable of blocking abnormal skin flaking processes characteristic of diseases such as atopic dermatitis. Monoclonal antibodies are proteins similar to those secreted by cells in the human immune system. They are produced in a laboratory for the diagnosis and treatment of various diseases.
Marcelo Zani, a postdoctoral researcher at the Federal University of São Paulo (UNIFESP) with a fellowship from FAPESP, is leading the project under the supervision of Vitor Oliveira, principal investigator at the CTS and based at the Center for the Study of Poisons and Venomous Animals (CEVAP) at the Botucatu campus of São Paulo State University (UNESP).
“Today, the main treatments for atopic dermatitis are corticosteroids and immunomodulators, which act on inflammatory response pathways and the immune system,” says Zani. “The main problem with these drugs is that continuous use isn’t recommended due to the various side effects related to modulating the inflammatory and immune responses, including the development of resistance by the patient,” he points out.
The diseases
Skin renewal is a natural bodily process, but it can become harmful in some pathologies. The process involves certain proteases, which are proteins that break down other proteins. These proteases act to transform the skin barrier. Disrupted functioning or a lack of natural inhibitors of these enzymes can trigger uncontrolled tissue desquamation.
Netherton syndrome, a rare condition, causes constant skin peeling accompanied by rashes, swelling, and hives. Atopic dermatitis, a less severe manifestation, causes dry skin and persistent itching, which can lead to wounds. According to the Brazilian Society of Dermatology (SBD), this condition affects 15% to 25% of children and 7% of adults in Brazil, and its incidence has increased in recent years.
Origin of the idea
While pursuing his doctorate at the Federal University of ABC (UFABC) in the laboratory of Professor Luciano Puzer, Zani began studying a new molecule with an innovative treatment proposal for the disease. The researchers developed a drug that can inhibit the enzyme associated with excessive skin shedding (tissue kallikrein 7, or KLK7), thereby preventing the manifestation of atopic dermatitis.
The drug emerged from UFABC’s research to discover new antibodies using a technology called phage display. Puzer learned about the methodology at a university in Germany and introduced it to the laboratory in Brazil. In this technique, viruses that infect bacteria (bacteriophages, or phages) are modified to display proteins, such as antibodies, on their surface. This display allows researchers to select phages with the greatest ability to bind to specific targets related to different diseases.
When Zani arrived at UNIFESP in Professor Oliveira’s Department of Biophysics, he brought the phage display technology with him. The technology was mainly applied to the study of antibodies with affinity for the virus that causes COVID-19. Due to the effectiveness and practicality of this methodology, the group expanded its use to other projects searching for antibodies and became involved in studies of a drug for treating atopic dermatitis.
How it works
According to Zani, the drug formulation combines a new recombinant antibody with a topical application route using a hydrogel. While there are products on the market that use recombinant antibodies to treat atopic dermatitis, they are systemic formulations that are administered via subcutaneous injection.
Therefore, this is the first proposal for antibody-based treatment via topical application, as it aims to inhibit an enzyme located in the most superficial layer of the skin. This allows for localized treatment, reducing the risk of side effects. Another benefit of local application is that smaller amounts of the active ingredient can be used, which reduces manufacturing costs.
Zani suggests that the drug may have other applications since the target enzyme is related to the development of other skin diseases such as psoriasis, a highly prevalent disease in Brazil and worldwide that causes red patches accompanied by intense itching or burning.
Development
The drug for atopic dermatitis is currently in the proof-of-concept stage. The goal is to evaluate the efficacy and safety of the proposed dosage. This phase is being carried out in partnership with several public and private research institutions and involves testing on animal models. In this case, the models are dogs with atopic dermatitis.
“The choice of dogs as a model was crucial for this stage, since these animals are naturally affected by atopic dermatitis, so the disease doesn’t need to be induced in the animal. In rodent models, where the disease is induced, there’s disruption of the inflammatory pathways in the animal, and since our drug doesn’t act on these pathways, such a model isn’t suitable,” Zani points out.
After undergoing efficacy and safety testing, the drug must undergo preclinical and clinical trials in humans, as well as drug characterization tests, which include stability testing. Only then can it be registered with regulatory agencies, such as the Brazilian Health Regulatory Agency (ANVISA), and become available in the healthcare system. The product already has a patent application registered in Brazil, the United States, and Europe.
It is the first bioproduct from Biotecnologia KRABS, a startup founded by Zani and Puzer. Professors Oliveira and Jair Chagas from UNIFESP and CTS-CEVAP are partners, and the FAPESP Program for Innovative Research in Small Businesses (PIPE) supports the company.
In future stages, the drug may be produced on a pilot scale for clinical trials at V-Biopharma, the Biopharmaceutical Teaching Factory linked to CTS-CEVAP and based at UNESP (read more at agencia.fapesp.br/52098). The facility allows production to be carried out in accordance with good manufacturing practices.
“That’s one of the main reasons we’re linked to CTS-CEVAP. Our bioproduct can be developed at V-Biopharma, as we need to maintain safety and reproducibility,” Oliveira points out.
* With information from Juliana Marques from CTS-CEVAP
Republish
The Agency FAPESP licenses news via Creative Commons (CC-BY-NC-ND) so that they can be republished free of charge and in a simple way by other digital or printed vehicles. Agência FAPESP must be credited as the source of the content being republished and the name of the reporter (if any) must be attributed. Using the HMTL button below allows compliance with these rules, detailed in Digital Republishing Policy FAPESP.





