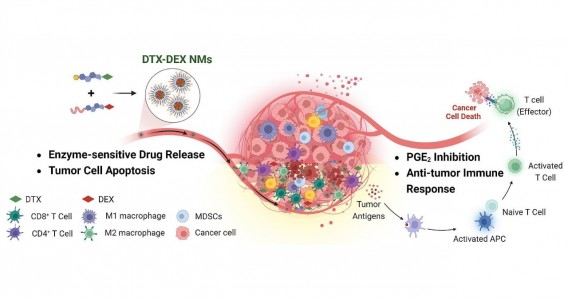
Em testes com animais, nanopartículas contendo substâncias já aprovadas para uso humano reduziram a inflamação no microambiente biológico em que cânceres desse tipo se instalam e vicejam, facilitando a ação do sistema imune
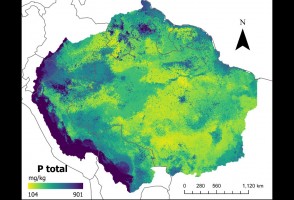
Mapas desenvolvidos com inteligência artificial confirmam baixos níveis de fósforo no solo da Amazônia
Pesquisa traz nova metodologia para descrever quantidade do mineral, que tem importância no ciclo de crescimento da vegetação e pode afetar a resposta da floresta às mudanças climáticas
Registro de produtos para controle biológico de pragas ultrapassa o de agroquímicos no Brasil
Cerca de 90% da área voltada ao cultivo de cana-de-açúcar no país hoje já utiliza inimigos naturais para o combate de pragas agrícolas; dados foram apresentados durante a FAPESP Week Illinois

Estudo focado na atenção primária busca melhorar diagnóstico e manejo da doença pulmonar obstrutiva crônica
Projeto conduzido por pesquisadores da USP e da Universidade de Birmingham foi um dos 15 apresentados em evento que celebrou a parceria entre a instituição britânica e a FAPESP. Ocasião também marcou o lançamento do University of Birmingham Brazil Institute, cuja missão é estreitar os laços de pesquisa com o Brasil
Chamada com UE apoiará pesquisas em ciência e engenharia de materiais
FAPESP lança edital em parceria com a M-ERA.NET, rede financiada pela União Europeia; pré-propostas serão recebidas até 14 de maio
Livro destaca a relevância das mulheres na construção do pensamento geográfico
Um dos capítulos, resultado de pesquisa apoiada pela FAPESP, aborda a geografia da América Ibérica do século 18 sob o olhar da mulher indígena
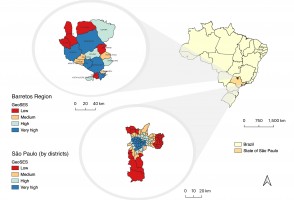
Maior acesso a exames eleva a incidência de câncer de tireoide em regiões paulistas mais ricas
Pesquisadores do Hospital de Amor e da Agência Internacional de Pesquisa em Câncer compararam a ocorrência geográfica e socioeconômica da doença na região de Barretos e na capital. Taxa de mortalidade foi similar entre as diferentes populações, o que, segundo os autores, sugere a ocorrência de diagnósticos em excesso em áreas de maior poder aquisitivo
OPORTUNIDADES FAPESP
PD em extensão rural
Feagri-Unicamp
Inscrições até 28/04/2024
Doutorado e mestrado em paleoceanografia e paleoclimatologia
IG-Unicamp
Inscrições até 30/04/2024
TT-4 em microscopia de força atômica
IQ-USP
Inscrições até 28/04/2024
PD em modelagem hidrológica e geoprocessamento
SemeAr/Embrapa Agricultura Digital
Inscrições até 28/04/2024
PD em ecologia aplicada e agroecologia
Esalq-USP
Inscrições até 28/04/2024
Chamada com UE apoiará pesquisas em ciência e engenharia de materiais
Livro destaca a relevância das mulheres na construção do pensamento geográfico
Registro de produtos para controle biológico de pragas ultrapassa o de agroquímicos no Brasil
Em palestra a empresários, ganhadores do Prêmio Nobel destacam a importância da pesquisa básica
FAPESP recebe propostas para novas Escolas de Ciência Avançada até 3 de maio
Obesidade abdominal associada à fraqueza muscular é condição que mais eleva risco de síndrome metabólica
USP apresenta sete novos centros de estudos ligados diretamente à reitoria
Cientistas transformam casca de banana em bioplástico para embalar alimentos
FAPESP e agência britânica vão apoiar projetos em ciências ambientais
Capítulo 6: Viagem ao rio Negro marcou reencontro após expedições pioneiras nos anos 1990

Uma das artes de pesca utilizadas para coletar peixes-elétricos na Expedição DEGy Rio Negro foi empregada pela primeira vez em larga escala em água doce no projeto Calhamazon, que reuniu pesquisadores do Brasil e dos Estados Unidos entre 1993 e 1996
Capítulo 5: Acidente com raia não atrapalha o ritmo das coletas no Jauaperi

Baixa gravidade da lesão, atendimento médico rápido e cuidados adequados fizeram com que pesquisador pudesse voltar aos trabalhos no mesmo dia em que foi ferroado por peixe peçonhento. Na Amazônia, casos muitas vezes se agravam por carência de assistência especializada
Capítulo 4: Sarapós se destacam pela diversidade de ambientes que ocupam

Os mais abundantes peixes-elétricos estão presentes desde o fundo dos grandes rios até os igarapés, onde podem se enterrar na areia ou se confundir com o folhiço. Em duas semanas, expedição na bacia do rio Negro coletou 27 espécies do grupo
Vídeos
Diário de Campo - Rio Negro
O Legado Suíço-Brasileiro na Amazônia: Arte, Ciência e Sustentabilidade
02/03/2024 a 30/04/2024
Introdução à programação com Python para alunas do ensino médio ou concluintes
30/03/2024 a 27/04/2024
Minicursos para estudantes do ensino médio 2024
06/04/2024 a 08/06/2024
Treinamento de plataformas e bases de dados científicas
09/04/2024 a 30/04/2024
14ª Semana da Computação
22/04/2024 a 26/04/2024
Esclarecimento da chamada de propostas para o CPE Citrosuco-FAPESP
23/04/2024 a 23/04/2024
Pulverização de lavouras vai ganhar aeronave especializada
Startup apoiada pelo PIPE-FAPESP está desenvolvendo helicóptero autônomo para pulverizar plantações em terreno íngreme
Teste irá permitir detecção pouco invasiva de micrometástases
Tecnologia pode possibilitar produção de enzimas no Brasil
Pulverização de lavouras vai ganhar aeronave especializada
Startup apoiada pelo PIPE-FAPESP está desenvolvendo helicóptero autônomo para pulverizar plantações em terreno íngreme
Chamadas FAPESP
Comunicar Ciência
Prazo: 22/01
Belmont Forum Climate, Environment, and Health
Prazo: Jan 2024
PIPE Start FAPESP-Sebrae: iniciando a jornada empreendedora de base tecnológica
Prazo: 18/03
Centros de Pesquisa em Inteligência Artificial Aplicada à Saúde
Prazo: 18/03
Fundação Nacional de Ciência da Suíça
Prazo: 22/03
Apoio a pesquisa em citricultura
Prazo: 31/03
Expedições Científicas Amazônia+10
Prazo: 29/04